Key takeaways
What Investors Should Watch:
- Pricing volatility is the new risk premium: When investors cannot confidently model U.S. pricing and reimbursement, discount rates rise and marginal programs fall below the funding line. Pricing uncertainty kills deals faster than revenue reduction.
- Geographic strategy signals pricing confidence: Companies launching U.S.-first while delaying Europe or Canada are protecting pricing integrity against international reference pricing. Watch for this strategic shift in launch sequencing as an early indicator of pricing pressure.
- Gross-to-net compression reveals market power: With intermediaries capturing nearly 50% of U.S. drug spending, manufacturers achieving sub-40% gross-to-net spreads demonstrate pricing discipline and formulary leverage — this metric matters more than list price.
- Early pricing integration separates winners from losers: Companies factoring value-based pricing into Phase 2 trial design (through endpoint selection, comparator choice, and health economics modeling) will command better valuations than those treating pricing as post-development.
- Non-dilutive capital flows are shifting: Early stage biotechs historically relied on ex-U.S. partnerships for validation and runway. U.S.-first strategies make these partnerships less attractive. Expect higher equity dilution and delayed European deals.
Where to Next:
- China emerges as the strategic wildcard: With U.S. pricing uncertainty rising, China’s evolution from manufacturing hub to innovation source creates competitive pressure on pricing power, IP strategy, and trial geography.
- Value thresholds are rising across all payers: Drugs addressing significant unmet needs (oncology, rare disease, gene therapies) will command returns, but only with demonstrable value over existing therapies. HTA readiness is no longer optional.
- Policy is reshaping capital allocation, not just revenue: Since the IRA passed, 55+ research programs halted and 26 medicines abandoned — track therapeutic area exposure to Medicare, MFN reference pricing, and negotiation timelines as core risk factors.
- The system must recalibrate globally: The U.S. has absorbed global R&D risk for decades through high pricing. As this dynamic changes, expect international price increases, delayed global access, or systemic innovation slowdown — model all three scenarios.
Introduction
The debate around drug pricing in the United States is often framed as a moral argument: Americans shouldn’t pay more than patients in other countries for the same medicines. Policies like the recently proposed Most Favored Nation (MFN) pricing and the previously introduced Inflation Reduction Act (IRA), were presented as mechanisms to relieve some of the imbalance. But the framing misses something fundamental.
Drug Pricing extends beyond simple economics ⎯ it reflects the value placed on healthcare innovation.
The past year marked a swift pivot in the macroeconomic environment. Trade policy shifts had broad and uneven effects across multiple sectors. Changes in the FDA, along with the discussion of Most Favored Nation policies led to uncertainty in the biotech space. Founders and investors want to understand how payer decisions impact innovation value.
Understanding drug pricing requires examining different payer systems and key stakeholders in the U.S. versus Health Technology Assessment (HTA) markets like Canada the UK, and some European countries.
The U.S. pricing landscape and its stakeholders
For decades, the United States has functioned as the profit engine underwriting global pharmaceutical innovation. It operates a multi-payer system divided into private and public payers, with the Centers for Medicare and Medicaid Services (CMS) as the dominant pharmaceutical spender. While other countries cap drug prices, the U.S. uses a market-driven but highly intermediated system. Multiple price benchmarks and reimbursement pathways exist, with patient costs varying by insurance coverage and negotiated terms.
Commercial players fall into several categories:
- Manufacturers that develop the drugs and set the list price and Wholesale Acquisition Cost (WAC)
- Managed Care Organizations (MCOs) are the health insurance companies that administer the health plans
- Pharmacy Benefit Managers (PBMs) are the industry’s middlemen, managing pharmacy benefits for MCOs, setting drug formularies and negotiating manufacturer rebates.
- Wholesalers manage the storage and distribution of medication
- Integrated Delivery Networks (IDNs) are the hospital systems that provide medical care to patients. They bill and submit reimbursement claims for pharmacy drugs administered within their sites-of-care.
- Pharmacies dispense medication to patients through mail-order, central fill, and retail locations.
All stakeholders except manufacturers are vertically integrated through informal relationships, mergers, acquisitions, or preferential contracts. The largest MCOs now control affiliated PBMs, dominating most U.S. prescription claims.
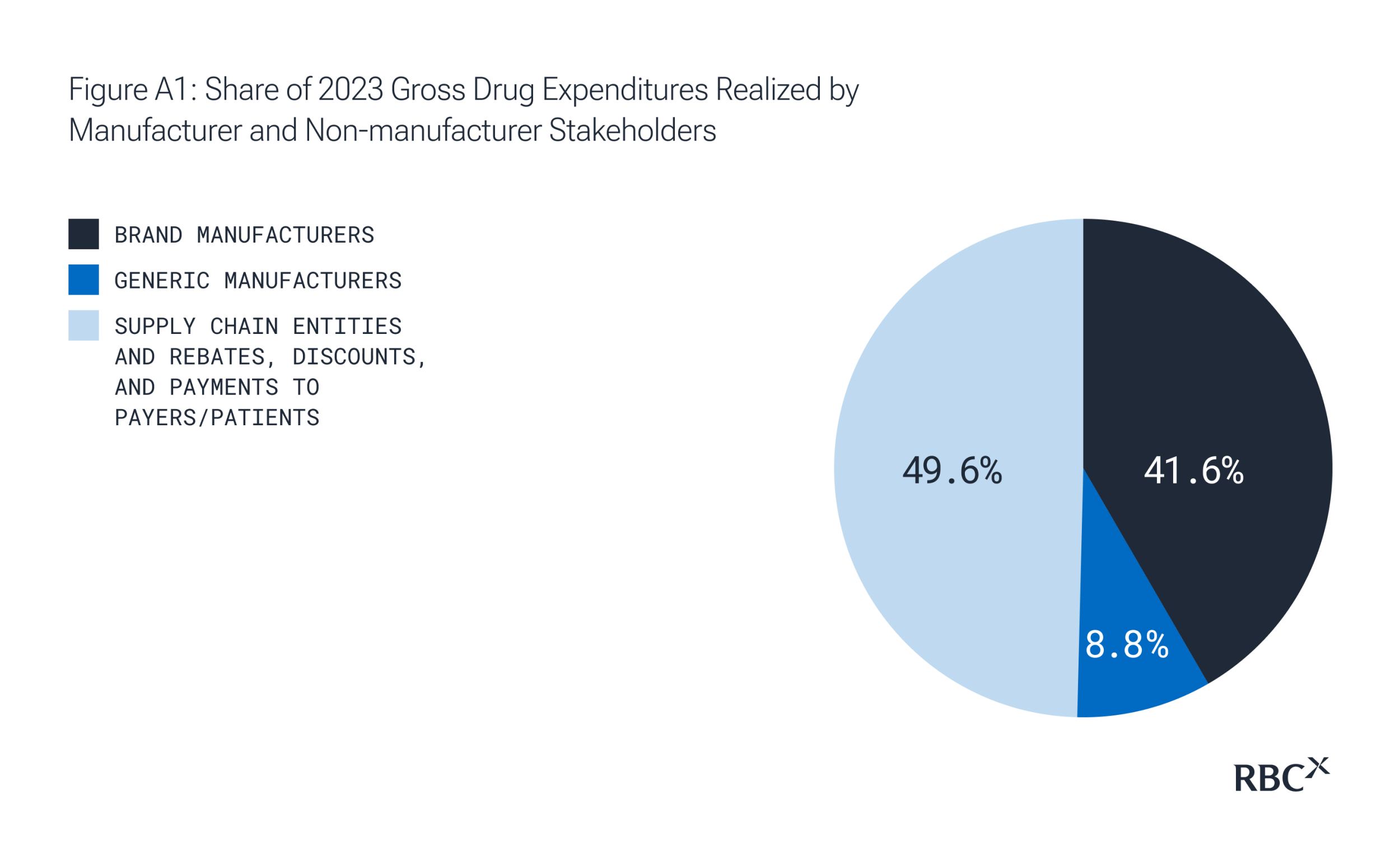
The multi-layered U.S. pharmaceutical supply chain redistributes nearly half of gross drug spending across this complex network of intermediaries before reaching patients, creating the pricing paradox.
The price the customer ends up paying
Before launch pricing, manufacturers conduct willingness-to-pay research with MCOs, PBMs, and IDNs to assess the current landscape, the appropriate price comparators, and contracting and rebate expectations. Manufacturers set and publish list prices, then negotiate preferred access with payers. Patient costs flow through multiple layers as illustrated:
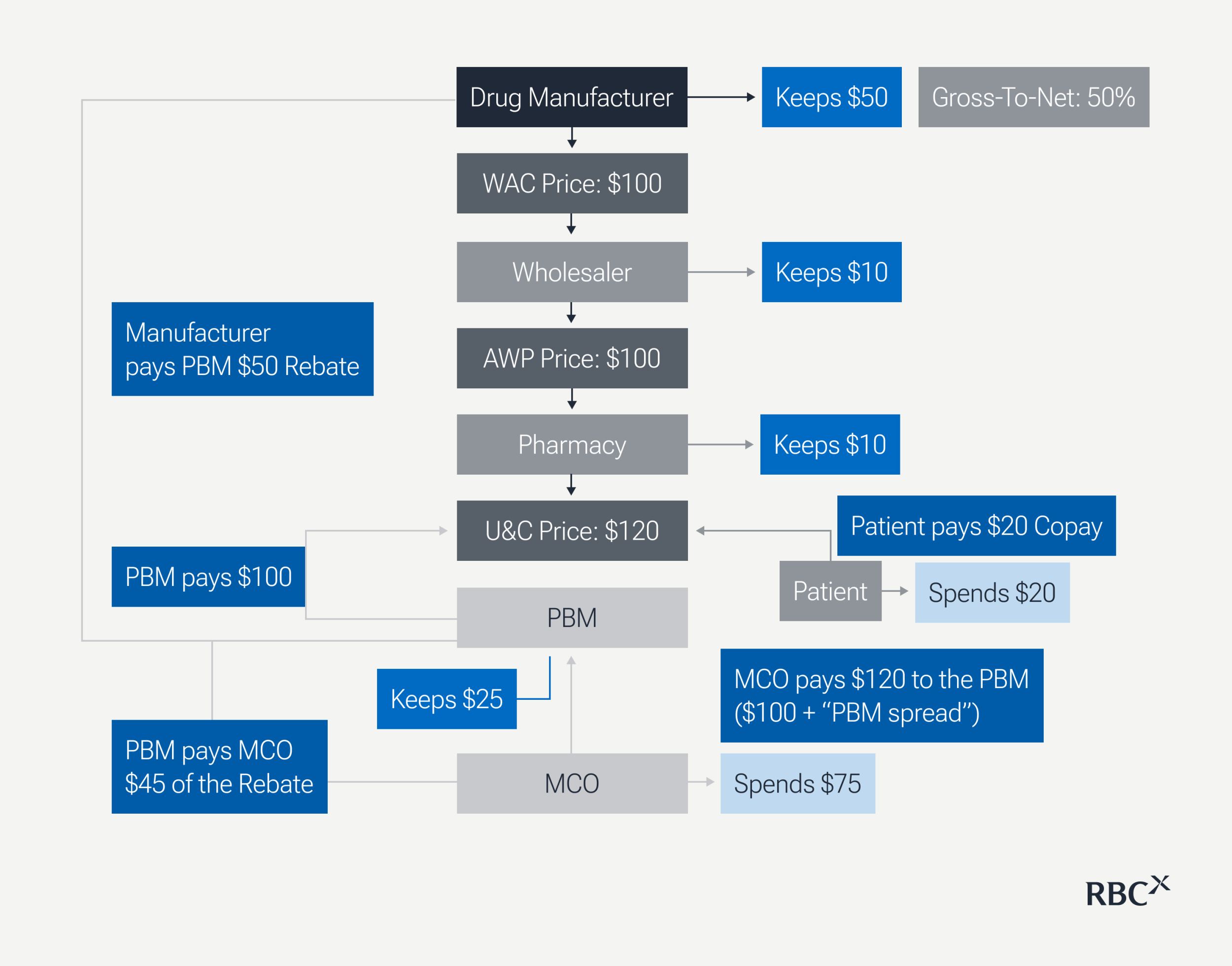
Consider a hypothetical drug with a $100 list price (WAC). This list price anchors the entire system ⎯ wholesaler markups, pharmacy reimbursement benchmarks, and rebate negotiations.
The Flow:
- Manufacturer sets WAC at $100 but pays a $50 rebate to the PBM for formulary placement, netting only $50 (50% gross-to-net reduction). Rebates strategically secure preferred status and prevent restrictions like prior authorizations.
- Wholesaler purchases at WAC, manages distribution and inventory, and earns a small spread ($10 in this example). It ensures supply chain availability and liquidity without influencing pricing.
- Pharmacy buys at Average Wholesale Price (AWP) of $110 (benchmark list price), dispenses to patients, receives $100 PBM reimbursement, and collects a $20 copay from the $120 usual & Customary (U&C) price — the standard retail “cash price” before insurance. U&C prices vary significantly by geography based on operating costs and local competition. This matters because uninsured patients pay the full U&C price, and insurers use geographic U&C benchmarks to set reimbursement rates and determine patient cost-sharing. The pharmacy’s $10 dispensing margin is PBM-dictated.
- PBM acts as the financial gatekeeper, controlling formulary access and manufacturer negotiation. It receives the $50 manufacturer rebate, passes $45 to the MCO (keeping $5), and retains the $20 spread between MCO charges ($120) and pharmacy reimbursement ($100), totaling $25 retained.
- MCO pays the PBM $120 upfront, receiving $45 in rebates, for a net cost of $75. It relies on PBMs to manage pharmacy benefits while maintaining competitive premiums.
- Patient pays a $20 copay based on inflated list price, not net price after rebates—a central policy concern. Uninsured patients (~8% of Americans) pay the full $120 U&C cost.
Tracing a single dollar through this system reveals the paradox: From the manufacturer’s $100 list price, only $50 reaches the innovator developing the drug. The other $50 flows through intermediaries — the PBM retains $25, the wholesaler $10, the pharmacy $10, and $5 covers various spreads. Meanwhile, the patient pays $20 based on the inflated list price, disconnected from the actual net cost. This fragmented system captures nearly 50% of drug expenditures through intermediation while simultaneously incentivizing higher list prices to accommodate all stakeholders. The result: manufacturers must price higher not just for innovation returns, but to fund the distribution system itself.
Pricing in the U.S. vs HTA markets
Unlike HTA markets, the U.S. imposes no explicit price ceiling at launch. Pricing remains market-driven despite aggressive payer negotiations. Net prices compress over time, but without upfront caps from a central authority.
Contrast that to a market like Canada:
- The Patented Medicines Pricing Review Board establishes a regulatory price ceiling.
- The Canadian Drug Agency (or INESS in Quebec) conducts HTA to assess value relative to existing treatments. In other words, if a manufacturer launches a drug in a therapeutic area that is already well served by inexpensive generics, they will need to show a significant improvement over the generic to get a significantly higher price approved.
- Reimbursement negotiations then begin with public drug plans through the pan-Canadian Pharmaceutical Alliance (pCPA) or private payers.
- Even after regulatory approval, provincial, territorial or national negotiations on pricing and reimbursement can take years.
The result is structurally lower prices due to regulatory ceilings and delayed access. Health Canada approvals lag FDA approvals by roughly two years. Investors understand this implicitly; Phase 2 models rely on U.S. peak sales and net pricing durability, not German or Canadian net price assumptions.
The uncomfortable truth: Most countries have benefited from higher U.S. pricing for decades. Lower international prices persist because U.S. margins absorb the R&D risk. Change this dynamic, and the entire system will need to recalibrate to find a new economic model.
MFN and IRA: The Real Question Is Not “Will Prices Fall?”
Much of the current discussion assumes Most Favored Nation (MFN) pricing or Inflation Reduction Act (IRA) negotiation will dramatically reduce drug prices in the U.S. The reality is more nuanced.
In May 2025, the U.S. administration signed an executive order mandating that Americans pay no more than the lowest reference country price (the MFN pricing). This was a measure separate from the Biden-era Inflation Reduction Act (IRA).
Introduced in 2022, the IRA capped Medicare out-of-pocket costs for prescription drugs at $2,000, shifting the liability for higher-cost drugs from patients to Medicare and manufacturers. Since the IRA passed, companies have halted at least 55 research programs and abandoned 26 medicines, raising questions around price controls and MFN policies impact on drug discovery.
Under the IRA Medicare Drug Price Negotiation Program, CMS sets Maximum Fair Prices (MFPs) with manufacturers. Non-compliance triggers severe fines or loss of Medicare and Medicaid market access.
The Trump administration is advancing MFN pricing initiatives to align U.S. drug costs with international benchmarks. CMS has proposed three new models: the Global Benchmark for Efficient Drug Pricing (GLOBE) for Part B drugs and the Guarding U.S. Medicare Against Rising Drug Costs (GUARD) model for Part D drugs — which would tie manufacturer rebates to international prices via mandatory Medicare demonstrations, and the Generating cost Reductions for U.S. Medicaid (GENEROUS)) which at this point is still voluntary. Still in proposal or pilot stages, these models aim to leverage global pricing data to reduce primarily Medicare and Medicaid drug costs. Impact will vary depending on therapeutic area, and Medicare exposure.
While not finalized, the greater disruption is not immediate revenue loss but strategic uncertainty. When investors cannot confidently model U.S. pricing and reimbursement, risk premiums rise, discount rates increase, return thresholds shift, and marginal programs fall below the funding line.
Pricing policy does not need to decrease revenues to alter capital formation. It only needs to increase perceived volatility.
Innovation Is Not Binary — It’s Marginal
Policymakers frame the debate as access versus innovation, but the reality is more nuanced. A $2 billion Phase 3 oncology program is funded when risk-adjusted returns clear hurdle rates, not for philosophical or altruistic reasons.
If expected U.S. pricing declines or becomes less predictable:
- Some indications will not be pursued
- Some Phase 2 programs will not be licensed
- Some platform bets will not be financed
- Some early partnerships will not be signed
Early-stage biotechs could face acute challenges. Historically, non-dilutive capital from ex-U.S. partnerships has provided validation and runway. If launch sequencing shifts toward U.S. first strategies — delaying Europe or Canada — those partnerships become less attractive. Co-development deals become distribution agreements, upfronts shrink, validation weakens, and equity dilution rises.
MFN may not directly reduce innovation. It may just alter the mechanisms that fund it.
The Launch Sequencing Shift
One underappreciated consequence of tighter U.S. pricing is geographic reprioritization. As U.S. pricing compresses toward international benchmarks, companies will protect pricing integrity through:
- Launching in the U.S. first
- Delaying Europe to avoid international reference price anchoring
- Being more selective about indication sequencing
- Designing trials to optimize for commercial endpoints earlier
Ironically, price-equalizing policies may widen access timeline divergence or raise costs for other markets.
Value Will Still Win — But Proof Will Be Harder
An important counterpoint is that drugs represent a minority of healthcare spending in developed systems. High-value therapies that address significant unmet needs (oncology, rare disease, transformative gene therapies) will continue to command meaningful returns.
Payers are not anti-innovation. Rather, they resist uncertainty and low-value spending. But the bar is rising. Value-based pricing is growing in biotech, and value varies by market.
For founders, pricing strategy cannot be a post-Phase 2 afterthought. It must inform:
- Indication selection
- Comparator choice
- Endpoint design
- Health economics modeling
- Market access strategy
The “science first, pricing later” era is over. Companies must integrate pricing strategy early to demonstrate value and stay competitive.
What Investors Should Be Watching
For investors, key signals are structural shifts, not political headlines:
- Are partnership upfronts declining?
- Are more companies raising dilutive equity earlier?
- Are the therapeutic areas more exposed to Medicare?
- Are Phase 3 trial designs becoming more payer-driven?
- Are non-U.S. launches being systematically delayed?
- Is capital concentrating in fewer, higher-certainty therapeutic areas?
If the answers trend toward yes, then pricing reform is already influencing capital allocation — even if headline prices appear stable.
Where to next?
None of this suggests the global drug pricing system is optimal. It is expensive, politically fraught, and structurally inefficient. But we should acknowledge the strategic trade-off that drug pricing policy isn’t about affordability alone. It is about who underwrites global biomedical risk.
If the U.S. reduces its willingness to pay at the margin, other countries must decide whether they are willing to assume more of that “burden”. With a perceived change in the whole economics of drug pricing, a likely outcome could be increased pricing in HTA markets.
With price uncertainty comes opportunity. China, once a manufacturing and generics hub, has become a major source of innovation. This shift impacts pricing power, trial strategy, IP, and global competition. The question is not whether pricing reform will affect biotech, it’s how much innovation capital the system is willing to reallocate or even relocate.
RELATED TOPICS
Other articles you may be interested in
